 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Zinc(II) and the ligand L form. a product cation that absorbs strongly at 600 nm. As l
ong as the concentration of L excess that of zinc (II) by a factor of 5, the absorbance of the solution is only lined on the cation concentration. Neither zinc(I) nor L absorbs at 600 nm. A solution that is 1.60X10-6mol·L-1in zinc (II) 1.00 mol·L-1in L has an absorbance of
0.164 in a 1.00 cm cell at 600 nm.Calculate
(1) the transmittance of this solution.
(2) the transmittance of this solution in a 3 cm cell.
(3) the molar absorbance of the complex at 600 nm.
 答案
答案





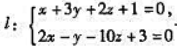 与平面II:4x-2y+z-2=0,则().
与平面II:4x-2y+z-2=0,则().






















